Digital Twins In Healthcare Market: Perspective, Comprehensive Analysis, Size, Share, Growth, Segment, Trends And Forecast, 2031

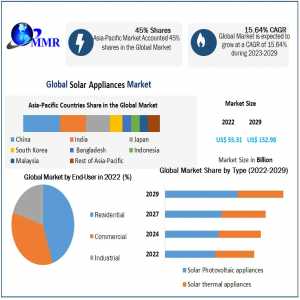
The Digital Twins in Healthcare Industry, valued at USD 610.1 million in 2023, is anticipated to experience exponential growth over the next several years. By 2031, the market is projected to reach USD 4504.73 million, expanding at an impressive compound annual growth rate (CAGR) of 25.97% from 2024 to 2031. Digital twins, virtual replicas of physical entities, are revolutionizing healthcare by enabling enhanced simulation, monitoring, and predictive analytics, thus improving patient outcomes and operational efficiencies.
Regional Outlook: The digital twins in healthcare market are seeing robust growth across various regions, including North America, Europe, Asia-Pacific, and the rest of the world. North America currently leads the market due to its advanced healthcare infrastructure, significant investments in healthcare IT, and strong presence of key market players. Europe follows closely, driven by increasing healthcare expenditure and the adoption of innovative medical technologies. The Asia-Pacific region is expected to witness the fastest growth, supported by rapidly evolving healthcare systems, increasing adoption of digital health technologies, and rising awareness about the benefits of digital twins in healthcare.
Key Growth Drivers of the Market: Several factors are propelling the growth of the digital twins in healthcare market, including:
-
Technological Advancements: Continuous innovations in AI, IoT, and big data analytics are enhancing the capabilities and applications of digital twins in healthcare.
-
Increasing Demand for Personalized Medicine: Digital twins enable highly personalized treatment plans by simulating individual patient responses, thus driving their adoption in precision medicine.
-
Improved Healthcare Outcomes: The ability of digital twins to provide predictive insights and real-time monitoring is improving patient care and operational efficiencies, boosting market growth.
-
Rising Healthcare Expenditure: Increased investments in healthcare infrastructure and digital health technologies are supporting the growth of the digital twins market.
Strengths of the Market: The digital twins in healthcare market have several inherent strengths:
-
Enhanced Predictive Analytics: Digital twins offer advanced predictive analytics capabilities, enabling early diagnosis and proactive treatment.
-
Operational Efficiency: These technologies improve hospital management and patient care processes, leading to better resource utilization and reduced costs.
-
Patient-Centered Care: Digital twins facilitate a more personalized approach to treatment, improving patient outcomes and satisfaction.
Impact of the Recession: Economic downturns can impact healthcare budgets; however, the potential for long-term cost savings and improved patient outcomes provided by digital twins can justify continued investment. The focus on efficiency and effectiveness in healthcare can help mitigate the impact of economic challenges on the digital twins market.
Key Objectives of the Market Research Report: A comprehensive market research report on the digital twins in healthcare market aims to:
-
Analyze Market Trends and Dynamics: Understand current trends, key drivers, and potential challenges shaping the market.
-
Evaluate Regional Market Performance: Assess growth opportunities and market dynamics across different regions.
-
Examine Competitive Landscape: Identify key players, their market strategies, and competitive positioning.
-
Forecast Market Growth: Provide detailed projections for market size and growth over the forecast period.
-
Identify Investment Opportunities: Highlight areas of potential investment and strategic growth.
In conclusion, the Digital Twins in Healthcare Market is set for remarkable growth, driven by technological advancements, increasing demand for personalized medicine, and the potential for improved healthcare outcomes. A detailed market research report can provide valuable insights for stakeholders aiming to capitalize on this dynamic and rapidly evolving market
Other Reports You May Like:
3D Cone Beam CT System Market Trends
Next Generation Sequencing Market Trends
About US:
SNS Insider has been a leader in data and analytics globally with its authentic consumer and market insights. The trust of our clients and business partners has always been at the center of who we are as a company. We are a business that leads the industry in innovation, and to support the success of our clients, our highly skilled engineers, consultants, and data scientists have consistently pushed the limits of the industry with innovative methodology and measuring technologies.
We assist our clients to anticipate industrial, economic, and consumer trends to drive disruptive change by fusing global experience with local information from experts throughout the world. We bring context to strategic and tactical data by bridging approaches based on data science and field research, assisting you in addressing your most pressing problems and spotting possibilities.
Contact Us:
Akash Anand – Head of Business Development Strategy,
info@snsinsider.com,
Phone: +1-415-230-0044 (US) | +91-7798602273 (IND),
Website: https://www.snsinsider.com
Author Bio
Article Comments
No Comments!
At present there are zero comments on this article.
Why not be the first to make a comment?
Similar Articles
Search Pages
User Upgrade
account to full use of editor,
Including hyperlinks
Article Categories
There are zero sub-categories in this parent category.
There are zero sub-categories in this parent category.